Clinical research sites rarely struggle with opportunity. What they struggle with is capacity. Clinical research coordinators (CRCs) are buried in regulatory binders, monitors occupy office space for days at a time, and the sheer volume of manual transcription creates a hard ceiling on growth. A site may have the investigators, the patient population, and even sponsor demand to run more trials, but the infrastructure cannot support it. Moreover, sponsors continue to increase trial volume, protocols grow more complex, and study teams are asked to manage more regulatory requirements, more patient data, and more monitoring oversight than ever before.
This is where the operating model begins to break down, and where eSource introduces a fundamentally different way of operating.
Today, many organizations are leveraging eSource, which not only improves adherence to the protocol, but begins to reposition site trial operations as an economic driver, enabling sites to convert capacity into measurable revenue growth.
By replacing a patchwork of manual systems with eSource – centralized, Part 11-compliant system – sites can reduce FDA audit risk, reclaim thousands of hours, and confidently expand their study capacity, driven by both increased study volume and the shift toward reimbursing site-controlled technology as part of trial delivery.
The Ceiling in Clinical Trial Site Operations
For savvy research organizations, eSource is fast becoming a capacity engine that enables sites to scale from managing a handful of trials to supporting significantly larger study portfolios.
In practice, the difference can look like this: A research site running 5 studies today may have the operational capacity to run 15 tomorrow. Not by hiring triple the staff, but by eliminating the operational inefficiencies that slow study execution.
Scaling efficiently requires fundamentally changing how your site captures, manages, and verifies data. In many organizations, source documentation still relies on:
- Paper binders or fragmented digital notes
- Manual transcription into EDC systems
- Sequential monitoring and verification workflows
Over time, this creates systemic drag that drag compounds:
- Coordinators spend the majority of their time documenting, not executing
- Data entry and reconciliation cycles slow timelines
- Monitoring becomes resource-intensive and disruptive
- Documentation variability increases operational risk
The outcome is predictable: sites reach a point where growth becomes constrained by staffing, and adding more studies requires adding more people.
The Multiplier Effect on Coordinator Capacity
When trial execution workflows become more efficient, the impact directly affects the number of studies a site can manage. For many research organizations, this operational shift produces a multiplier effect. The same team can support more patients, more visits, and more studies, without proportional increases in staffing.
What changes in practice:
- Coordinators spend less time on manual documentation
- Data is cleaner at first capture, reducing query cycles
- Monitoring becomes faster and less disruptive
- Teams can support more concurrent trials
Instead of hiring additional coordinators for every new study, sites can scale their research programs using more efficient workflows. Effectively, sites often move from managing a handful of studies to supporting significantly larger portfolios.
The Mechanics of Scale using eSource
The capacity gains enabled by eSource are rooted in specific workflow improvements.
- Elimination of transcription as a system requirement. Data captured once, at the point of care, removes the need for downstream re-entry and reduces the primary source of data discrepancies.
- Compression of data timelines. Real-time validation and structured capture reduce the time between visit, entry, and review, thereby accelerating the entire data lifecycle.
- Decentralization of monitoring. Remote access to source enables continuous oversight, which in turn reduces reliance on on-site visits, freeing both site and sponsor resources.
Individually, these improvements save time. Collectively, they create capacity. What emerges is not just a technology upgrade, but an operating model shift.
Traditional model:
- Documentation is manual, often captured in paper binders or fragmented digital notes
- Data must be re-entered into EDC systems, introducing duplication and error risk
- Monitoring is on-site and episodic, slowing review cycles and disrupting site operations
- Execution varies across coordinators and studies, increasing operational complexity
eSource-enabled model:
- Documentation is structured and captured in real time at the point of care
- Data is entered once and flows across systems without rework or transcription
- Monitoring becomes remote and continuous, enabling faster oversight and query resolution
- Execution is standardized across studies, improving consistency and performance
This is the difference between working harder and building a system that allows you to scale.
eSource: From Site Capacity to Site Growth
Capacity alone does not drive growth, but it enables it. As sites expand their operational capacity, they begin to see measurable shifts:
- Ability to accept additional studies without staffing increases
- Improved performance metrics (data quality and query resolution)
- Greater attractiveness to sponsors and CROs
This is where eSource moves beyond efficiency and becomes a growth enabler. More importantly, it begins to shift from a cost center to a revenue-generating capability, expanding both the volume of studies and the economic output of the site.
eSource: From Site Growth to Site Revenue Expansion
The impact of eSource extends directly to site economics. Leading research organizations are increasingly positioning eSource as a billable capability, not just an internal tool.
Findings from RealTime Reports – Site Pricing Strategies Survey Summary:
- Over 80% of respondent sites charge sponsors for eSource-related services
- More than half charge at levels that exceed their underlying costs
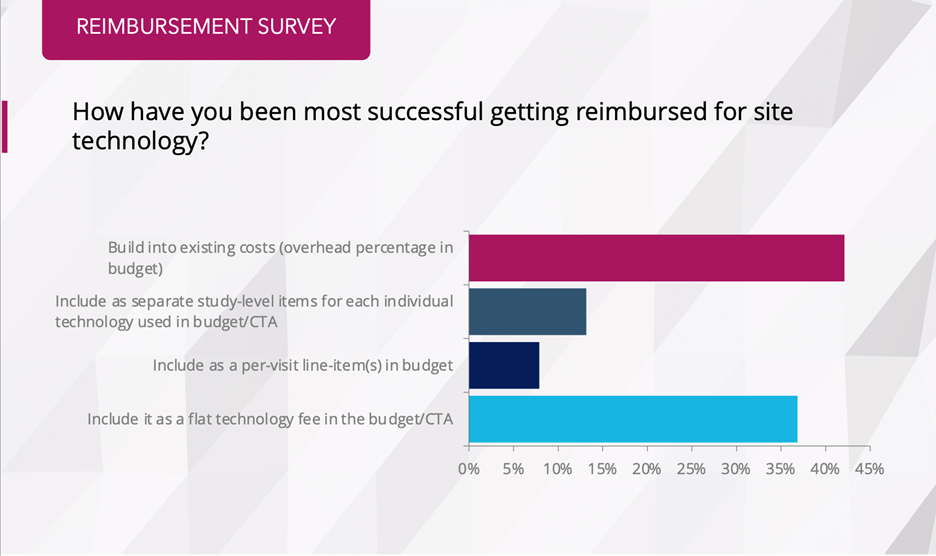
This reflects a broader shift in sponsor expectations. Sponsors are placing greater value on sites that can deliver:
- Structured, audit-ready data at the point of care
- Faster data availability
- Reduced monitoring burden through remote access
In this context, eSource becomes part of the value delivered to the sponsor, and part of the financial model of the site. The same infrastructure that expands capacity can also generate margin.
The BYOT Shift: Why Sites Now Control Their Infrastructure
At the same time, the industry is undergoing a structural shift in how technology decisions are made: Bring Your Own Technology (BYOT). Historically, sponsors dictated the technology stack used in clinical trials, often requiring sites to adopt sponsor-selected systems for data capture and documentation.
That model is changing. Sponsors and CROs are increasingly:
- Allowing sites to operate within their own validated systems
- Prioritizing speed, data quality, and execution over rigid technology standardization
- Recognizing that site performance improves when workflows are consistent across studies
This shift has important implications.
For the first time, sites have greater control over their operational infrastructure, and greater accountability for performance. High-performing organizations are using this flexibility to standardize on platforms that:
- Reduce variability across studies
- Improve coordinator efficiency
- Enable consistent, scalable execution
eSource is becoming a foundational component of that infrastructure. Within a BYOT model, eSource is becoming part of the site’s core operating environment, applied consistently across sponsors, protocols, and therapeutic areas.
The Bottom Line
Sponsors are increasingly selecting sites based on performance speed, data quality, and reliability. Technology infrastructure is now a signal of that capability. Sites that invest in modern, standardized, and scalable workflows position themselves as:
- Lower risk
- Higher performance
- More scalable partners
In multi-site networks and health systems, this becomes even more critical, where consistency and visibility across studies determine overall performance.
eSource is often introduced as a way to replace paper. That framing is too narrow. At scale, eSource does four things simultaneously:
- Expands coordinator capacity
- Enables portfolio growth
- Supports new revenue models through sponsor reimbursement
- Reduces audit risk and protocol deviations through structured, real-time workflows
eSource doesn’t just reduce cost. It changes the economics of running a research site, unlocking a more scalable, more profitable operating model.
Talk to an expert to see the platform in action.
Read More: RealTime Reports: Negotiating Sponsor Reimbursement for Site-based Technology
Read More: What’s Broken in Clinical Trial Oversight?
FAQs
Q: What is eSource in clinical trials?
A: eSource refers to the electronic capture of clinical trial data at the point of care, replacing paper-based or transcribed source documentation. Instead of recording data manually and re-entering it into other systems, data is captured once, in real time, within a compliant digital environment.
Q: How does eSource increase site capacity?
A: eSource increases capacity by removing the biggest operational bottlenecks—manual documentation, transcription, and delayed monitoring. By capturing data once and enabling real-time validation and remote review, coordinators spend more time executing trials and less time managing paperwork, allowing the same team to support more studies.
Q: Does eSource actually reduce the need for additional staff?
A: In many cases, yes. Rather than scaling linearly with headcount, sites using eSource scale through workflow efficiency. This means organizations can take on additional studies without hiring proportionally more coordinators, especially as documentation and monitoring processes become more streamlined.
Q: How does eSource impact data quality?
A: eSource improves data quality by capturing structured data at the point of care and reducing transcription errors. Real-time validation ensures cleaner data upfront, which leads to fewer queries, faster resolution cycles, and more reliable datasets for sponsors and CROs.
Q: Can eSource reduce FDA audit risk?
Yes. eSource systems that are 21 CFR Part 11 compliant provide audit trails, version control, and standardized workflows. This reduces variability in documentation and helps ensure that data is consistent, traceable, and inspection-ready at all times.
Q: How does eSource support remote monitoring?
Because data is digitized and accessible in real time, monitors can review source data remotely rather than relying solely on on-site visits. This enables continuous oversight, faster query resolution, and reduced disruption to site operations.
Q: Is eSource only valuable for large site networks?
A: No. While large networks see amplified benefits, even smaller sites can significantly increase their capacity and efficiency. eSource allows smaller organizations to compete with larger networks by improving execution speed, data quality, and scalability.
Q: How does eSource contribute to revenue growth?
A: eSource contributes to revenue in two ways:
- Increased capacity: Sites can run more studies without proportional cost increases
- Sponsor reimbursement: Many sites now charge for eSource-related services, turning technology into a billable capability rather than a cost center
Q: What is the BYOT (Bring Your Own Technology) model, and why does it matter?
A: BYOT is a growing industry shift where sponsors allow sites to use their own validated technology rather than mandating sponsor-selected systems. This gives sites more control over their workflows, enabling consistency across studies and improving overall performance.
Q: How is eSource different from traditional documentation methods?
A: Traditional models rely on manual documentation, duplicate data entry, and on-site monitoring. eSource replaces this with real-time, structured data capture, single-entry workflows, and remote monitoring—transforming how trials are executed, not just how data is stored.
Q: Is eSource difficult to implement or adopt?
A: Modern eSource platforms are designed to align with existing clinical workflows, making adoption easier than legacy systems. When implemented correctly, sites often see rapid efficiency gains and strong staff adoption because the system reduces their workload.
Q: How does eSource make sites more attractive to sponsors?
A: Sponsors increasingly prioritize sites that can deliver:
- Faster data availability
- Higher data quality
- Reduced monitoring burden
eSource-enabled sites signal operational maturity and lower execution risk, making them more competitive during site selection.
Q: Is eSource just a replacement for paper?
A: No—and this is the critical misconception. While eSource replaces paper, its real value is in transforming the operating model. It standardizes execution, enables scalability, reduces risk, and creates new revenue opportunities.
Q: What kind of ROI can sites expect from eSource?
A: ROI typically comes from:
- Time savings (reduced documentation and data entry)
- Reduced monitoring and audit-related costs (faster, cleaner data)
- Increased study volume without proportional staffing
- Sponsor reimbursement for technology use
Together, these factors shift eSource from a cost-saving tool to a revenue-generating infrastructure.

