As eSource (electronic source) adoption in clinical trials increases, organizations looking to make the switch are also evaluating the cost-effectiveness and benefits of eSource over traditional data collection methods. eSource enables clinical trial sites to capture, manage, and store data digitally, streamlining workflows and enhancing data accuracy. In contrast, traditional data collection methods, including paper case report forms (CRFs), spreadsheets, and manual data entry, have been the standard for decades but come with limitations that impact efficiency, cost, and data integrity. This blog will dive into a cost-benefit analysis of eSource versus traditional data collection methods in clinical trials, focusing on long-term savings, efficiency gains, and the return on investment (ROI) associated with adopting eSource.
Cost Analysis of eSource
Initial Investment Costs
The initial costs associated with eSource implementation include purchasing or licensing eSource software, training staff, and potentially upgrading technological infrastructure to support digital data capture and secure storage. However, this upfront investment is offset by long-term savings as operational efficiencies increase and the need for paper, storage, and manual labor decreases.
In contrast, traditional data collection methods have lower immediate costs, as most sites already have the infrastructure and staff required to handle paper documentation and data entry. However, the lack of efficiency and high potential for errors mean that long-term costs may be higher due to the labor-intensive nature of these methods, storage requirements, and costs associated with error reconciliation.
Labor and Operational Costs
Labor costs are one of the most significant differences between eSource and traditional data collection methods. With eSource, data collection is streamlined, eliminating the need for manual data entry and reducing time spent on error correction. Built-in validation checks in eSource systems catch inconsistencies at the point of entry, minimizing the need for rework and oversight. This reduction in labor allows clinical trial staff to focus on higher-value activities, such as patient care, study management, and quality assurance, rather than on repetitive data entry and correction.

On the other hand, traditional data collection methods require dedicated staff for data transcription, data entry, and document management. In addition, each step in the process – from collecting data on paper to manually entering it into a database –introduces potential for error, which can lead to further time and labor for reconciliation and verification. These processes can drive up costs significantly, particularly for large, multi-site trials.
Monitoring and Compliance Costs
Compliance is critical in clinical trials, and monitoring costs are often a substantial portion of a study’s budget. Traditional data collection methods typically require frequent on-site monitoring visits to ensure that data is collected accurately, transcribed properly, and compliant with regulatory standards. Travel expenses, staff time for data verification, and follow-up visits can add up quickly, especially in large or geographically dispersed studies.
eSource supports remote monitoring, enabling monitors to access trial data in real-time from any location. This capability reduces the need for on-site visits, which in turn lowers travel expenses, monitoring fees, and logistical costs. Additionally, eSource incorporates automated audit trails, which provide a transparent record of all data changes and actions taken. This level of detail simplifies the audit process, improves compliance, and decreases the risk of regulatory penalties, further reducing the overall monitoring costs.
Efficiency Gains Using eSource in Clinical Trials
Streamlined Data Collection and Real-Time Access
One of the primary advantages of eSource is the ability to streamline data collection processes. By capturing data directly in a centralized digital platform, eSource eliminates the need for manual data entry and the inherent risk of transcription errors. Real-time access to data allows researchers, sponsors, and monitors to view updated information instantly, facilitating faster decision-making and reducing delays in trial progress.
In contrast, traditional methods require multiple steps to enter and verify data, leading to delays that can slow down study timelines. By cutting down on these manual processes, eSource accelerates data availability and enhances data quality, which can be crucial in time-sensitive studies.
Better Data Integrity
Data integrity is paramount in clinical trials, as data discrepancies can compromise study outcomes and even lead to regulatory non-compliance. eSource systems offer built-in validation checks that flag inconsistencies or errors at the point of entry, helping to maintain high data quality standards. This proactive approach to data accuracy reduces the need for costly corrections and rework later in the trial process.
Traditional data collection methods, however, do not have these validation features. Human error during data entry can lead to inaccuracies, and without automated checks, discrepancies may go unnoticed until later stages, potentially jeopardizing trial results. With eSource, data quality is maintained throughout the study, reducing both time and costs associated with data correction.
Return on Investment (ROI): eSource as a Profit Center
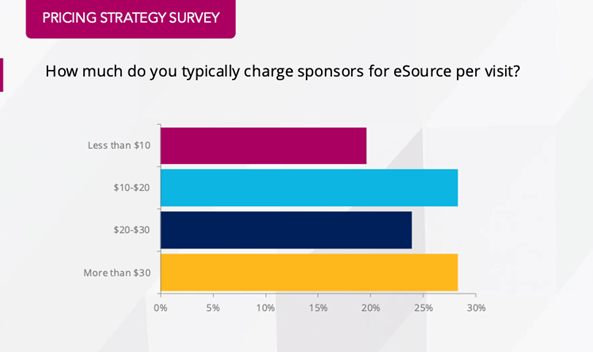
According to the RealTime Reports: Clinical Research Site Pricing Strategy, eSource is not only reducing operational costs but also generating positive revenue for many sites. The RealTime Report shows that over 80% of sites are charging more than their costs for eSource services, turning eSource into a profit center. In fact, more than half of these sites are charging double or even triple their costs, significantly boosting their bottom line.
Final Thoughts
eSource offers a clear financial and operational advantage over traditional data collection methods. Although eSource requires an upfront investment, the long-term financial benefits are considerable. The reduction in labor costs, fewer on-site monitoring visits, increased compliance efficiency, and improved data integrity all contribute to a positive ROI over time. By minimizing operational and monitoring expenses, organizations using eSource can often see a return on investment within the first few trials, as they save on personnel hours, reduce delays, and improve trial accuracy.
Traditional methods, while lower in initial cost, offer little in terms of long-term ROI. The ongoing labor, storage, and error correction costs add up over time, diminishing the financial efficiency of paper-based approaches. For organizations looking to reduce overhead and enhance productivity, eSource presents a compelling case for investment, yielding greater savings and supporting scalable growth.
Case Study: RealTime-eSource: Fueling 28x Revenue Growth at the University of the Sunshine Coast
Read More: eSource vs EHR/EMR – Why Sites use eSource for Clinical Trials

